say: daw-noe-ROO-bi-sin
The Chem Abstracts (CA) name for this compound is: (8S-cis)-8-acetyl-10-[(3-amino-2,3,6-trideoxy-alpha-L-lyxo-hexopyranosyl)oxy]7,8,9,10-tetrahydro- 6,8,11-trihydroxy-1-methoxy-5,12-naphthacenedione. You can see why people like to call it something simple like daunorubicin! Other names by which you might hear it called are daunomycin, leukaemomycin C, rubidomycin, or Cerubidin. They all refer to the same compound, which has the CA registry number 20830-81-3.
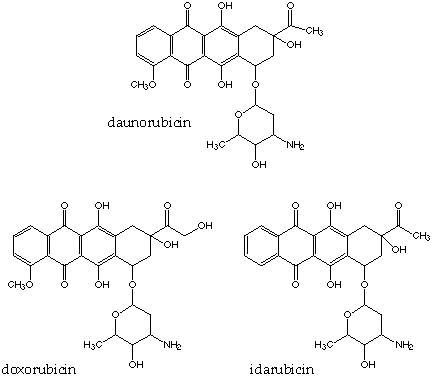
Daunorubicin is an anthracycline antibiotic, as is idarubicin (CAS 58957-92-9) and doxorubicin or adriamycin (CAS adriamycin 23214-92-8). Anthracycline antibiotics were first isolated as red substances from microorganisms in 1939, and their antibiotic properties were studied in the 1950s. These antibiotics killed bacteria quite readily, but were too toxic to be used for infections in humans. It wasn't until the 1960s that anthracycline antibiotics were tested for antitumor properties and found to be active against cancer cells.
Pure daunorubicin is a solid melting at 208-209 degrees centigrade. The LD50 in mice is 50 mg/kg of body weight. Most of the information I found about this group of drugs was specific for daunorubicin or doxorubicin. References allude to the same side effects in each anthracycline drug, some more, some less, and different anthracyclines work better against different cancers. Daunorubicin is more effective against leukemia cells than solid tumor cells.
Daunorubicin works to prevent cell growth by preventing DNA replication. The exact mechanism of this effect is still being studied by chemists and molecular biologists. One theory is that it is a topoisomerase inhibitor. In order to be replicated, the DNA double helix must be first pulled apart. Topoisomerase enzymes accomplish this by temporarily cleaving one strand of the DNA and passing the other strand through the break, then reattaching the cut ends. Daunorubicin prevents the topoisomerase from reattaching the broken ends. Studies have shown that anthracycline antibiotics intercalate into the double strands of DNA; this could disrupt the DNA and thus DNA replication. Anthracyclines also cause free radical breakage of the DNA in themselves. Thus, in replicating cells numerous DNA strand breaks appear and the cells die.
Research from the lab of Tad Koch, including Dylan Taatjes, a graduate student (now a professor) in the chemistry department at CU Boulder provides more detail on the mechanism of action of doxo- and daunorubicin. In the cell, the quinone portion of the drug molecules catalyze the production of formaldehyde in a redox reaction sequence. Formaldehyde in turn reacts with the amino-sugar portion of the molecule to form what they propose is the active forms of the drugs. The active form of the drug intercalates into the DNA, forming a virtual cross-link and alkylating the DNA. They treated doxo- and daunorubicin with formaldehyde and studied the interaction of the resulting doxo- and daunoforms with DNA in aqueous medium and the cytotoxicity of these compounds on cancer cells. They found them up to10000 times more cytotoxic. Presumably this is because they skip the formaldehyde activation step in the cell. Also, by skipping the redox reaction, they expect doxo- and daunoform to be especially effective in chemotherapy resistant cancer cells, because the resistant cells have decreased redox capabilities, and thus cannot form the active form of doxo- and daunorubicin. Also, resistant cells over express a particular glycoprotein that serves to pump all the doxo- and daunorubicin out of the cells. The doxo- and daunoforms are more lipophilic in character and therefore are not as good as substrates for the glycoprotein and are retained in the cancer cells to a larger degree.
3/98: Another promising anthracycline - Epidoxoform. This compound is the formaldehyde conjugate of epidoxorubicin which is the 4'-epimer of doxorubicin. In other words, it is a dimer formed by formaldehyde treatment like doxo- and daunoform, but they begin with a compound that has an HO- group pointing in the other way than it does in doxorubicin. Epidoxorubicin has been approved for human use in all countries except the U.S. Epidoxoform has a different structure than Doxoform because of this change in stereochemistry at the 4'-position. A major consequence of the structural change is a higher level of hydrolytic stability. Although it is less active than Doxoform against cancer cells, its stability has led us to use it in the first mouse experiments being conducted at CSU school of veterinary medicine.
Daunorubicin can also sometimes cause mutations in normal cells. A person treated with daunorubicin is at higher risk for certain cancers throughout his/her lifetime. It also causes sores and ulcerations if it escapes the vein.
Long-term anthracycline therapy and a cumulative dose of about 550 mg/square meter (!) causes an unusual heart toxicity. These drugs kill cardiac myocytes, and if enough myocytes are lost the heart muscle fails. This is an irreversible process. (More on late effects of the anthracyclines is available on the ped-onc-survivors web site.)
Scientists are constantly studying daunorubicin and other anthracycline derivatives. They are trying to find a derivative that is more specific to tumor cells. Another strategy is to "wrap" them in liposomes to protect normal cells from the toxic effects. A new liposomal version of doxorubicin called Doxil has been recently approved as a Kaposi's sarcoma treatment.
Daunorubicin is given by IV. It is orange in color when in solution. Side effects that usually do not require medical attention are:
Anything else should be reported to your oncodoc. Another sheet we received from the hospital says:
"The possible adverse effects of this drug are severe nausea, vomiting, mouth ulcers, and depression of the bone marrow, which leads to a depression of the white cells that are important for fighting infection, depression of blood platelets which are important in blood clotting, and depression of red blood cells which transport oxygen. Other adverse effects are hair loss, local irritation and necrosis if the medication escapes the vein. This drug in a high enough dose can produce changes in the rate and rhythm of the heart beats by an effect on the heart muscle itself. This drug has an unusual property, namely an accumulation effect. In other words, the body seems to accumulate toxicity regardless of how long it has been since receiving the drug previously. Therefore, a total lifetime dose has been determined which will not be exceeded by your doctors."
Still more, for a sheet given to us by the hospital, click here.
Zinecard (dexrazoxane) can be given during anthracycline treatment to protect the heart. See my discussion on the ped-onc resource center.
References: