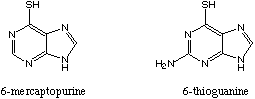
say: 6-mer-cap-toe-PURE-eene
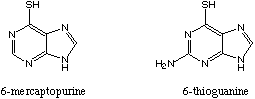
6-Mercaptopuring is commonly referred to by docs and pharmacists as purinethol or the simple and easy to say 6-MP. 6-mercaptopurine has the CAS Registry Number 50-44-2. The CA name is: 1,7-dihydro-6H-purine-6-thione. Trade names include Leukerin, Mercaleukin, Puri-Nethol, Purinethol.
When isolated from aqueous solution, 6-MP forms yellow prisms of a monohydrate form, becoming anhydrous at 140 degrees centigrade and decomposing at 313-314 degrees centigrade. The LD50 in mice and hamsters is 157 mg/kg of body weight. It is listed as an antineoplastic and immunosuppressant. The Merck Index lists two relatively recent review articles (1988 and 1991) of its use in the treatment of leukemia, if you are interested.
6-MP is a DNA antimetabolite. This means that it mimics a substance necessary in DNA synthesis necessary.
Digging up an old biochem text, I found the following information. In the body, 6-MP is converted to the corresponding ribonucleotide. 6-MP ribonucleotide is a potent inhibitor of the conversion of a compound called inosinic acid to adenylic acid. Without adenylic acid, DNA cannot be synthesized. [DNA is made up of A (adenine), T (thymine), G (guanine), and C (cytidine), each of which is linked in turn to deoxyribose, a sugar]. It also inhibits the conversion of inosinic acid to xanthylic acid that is necessary for the synthesis of guanylic acid. A different text states that 6-MP also works by being incorporated into nucleic acids as thioguanosine, rendering the resulting nucleic acids (DNA, RNA) unable to direct proper protein synthesis.
Cancer Growth and Progression, Volume 10, Cancer Management in Man, written in 1989, validates these two mechanisms of action of 6-MP: (1) inhibition of purine biosynthesis and (2) incorporation into DNA. This is a good reference for detail on the mechanism of purine antimetabolites, 6-MP and 6-thioguanine. These are the only two purine antimetabolites that are currently used in chemotherapy [1997]. However, there are many experimental antipurines being tested as anticancer drugs.
6-MP can be administered either in tablet form (orally) or IV. Common side effects listed on the sheet we received from the hospital are:
During administration, the oncodocs will continue to monitor white blood cell counts and liver function. In our experience, 6-MP is one of the less nasty chemo drugs.
This is the only sheet we got at the hospital for 6-MP.
TPMT and 6MP. Some people have different forms of TPMT (thiopurine methyltranserase), the enzyme that metabolizes 6MP (and 6TG). In fact, 1 in 300 individuals cannot break down 6MP at all. Some oncologists advise patients to be tested for TPMT activity before any dose of 6MP is administered; some doctors do not. There is a chance that the 6MP will cause the ANC to plummet so far that the patient will die from infection. I advise you to read the article in the CCCF Fall 2003 quarterly journal:
6-MP should be taken at night. According to an article on PostGraduate Medicine Online, this drug should be taken at night. "The study found that the risk of relapse was 2.56 times higher in children who received chemotherapy in the morning than in those receiving the same treatment in the evening."
6-MP and milk/foods. Many (but not all) parents are told by their oncologists to give 6-MP on an empty stomach, and never to give it at the same time as milk. The first link is to a MedLine abstract that discusses this topic, the second is to an article on the Pediatric Pharmacotherapy web site.
6-MP and allopurinol. 6-MP is a substrate for xanthine oxidase (XO) which converts it to 6-thiouric acid by oxidation. This appears to be an important route for inactivation of the drug. Allopurinol, a drug used to prevent the hyperuricemia and uricosuria that often follow marked cell kill consequent to leukemia therapy, inhibits this conversion. When 6-MP and allopurinol are used together, the dose of 6-MP is usually lowered. Notably, XO is found in high concentration in cow's milk, thus explaining why 6-MP should not be taken with milk.
6-MP/6-TG metabolites levels. The drug dose is determined by body surface area, but not all kids have the same levels of metabolites in their bloodstream after treatment. There is some evidence that this is an important factor.
Gertrude Elion synthesized and co-developed two of the first successful drugs for the treatment of leukemia (thioguanine and mercaptopurine). Read the fascinating article below:
Some abstracts on 6MP and 6TG, on the K.O.A.L.A. site. (Excellent resource.)Designing Drugs: Gertrude Elion, on Invention and Technology
say: 6-thigh-oh-GWAN-een
6-Thioguanine is closely related to 6-mercaptopurine, both in structure (see above) and in metabolism. Like 6-MP, 6-thioguanine is a purine antimetabolite. Thioguanine inhibits purine biosynthesis as does 6-MP, albeit at different steps: 6-TG is an analogue of huanine while 6-MP is an analogue of hypoxanthine. It is also incorporated into the DNA. (Please see the above discussion for 6-MP.)
Synonyms: 2-Aminopurine-6-thiol; Lanvis; Tabloid; TG; Wellcome U3B; X 27; 2-Amino 6MP; 2-Amino-6-mercaptopurine; 2-Amino-6-purinethiol; 2-Aminopurine-6(1H)-thione; 6-Mercapto-2-aminopurine; 6-Mercaptoguanine; 2-amino-1,7-dihydro-6H-purine-6-thione; THG; 6-TG. It is a pale yellow crystalline powder with a melting point greater than 360 degrees centigrade. The CA RN for 6-thioguanine is 154-42-7.
In our protocol (CCG 1961), 6-TG and 6-MP are used at different times for maintenance. I do not know the reason for this, except to assume that past studies found it effective. Historically, these are some of the older chemotherapy drugs, having been used for about 35 years. Both these drugs are given orally on arm B of CCG 1961. In general, 6TG shows greater liver toxicity. (A lot more information on the advantages/disadvantages of these two drugs is available; I'd advise a search of MedLine, the articles on the KOALA site, and Pui's book Childhood Leukemia if you are interested.)
Possible side effects:
Less common side effects: